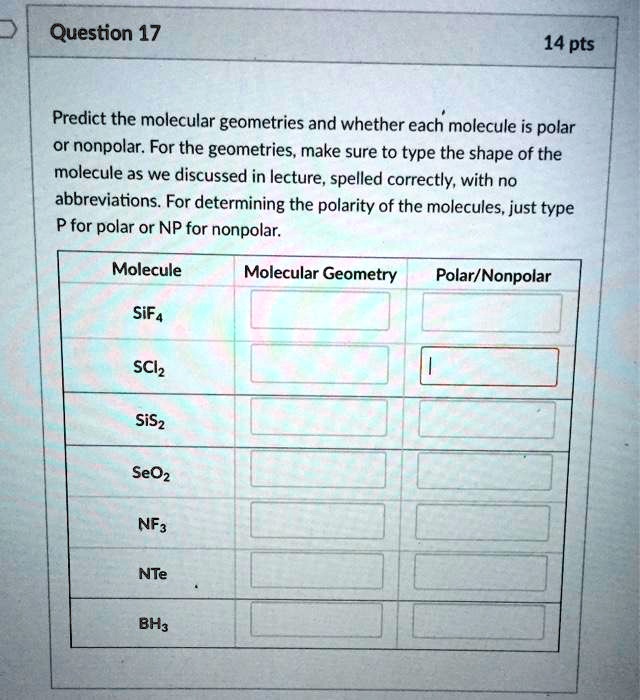
SOLVED: Question 17 14 pts Predict the molecular geometries and whether each molecule is polar nonpolar: For the geometries, make sure to type the shape of the molecule as we discussed in
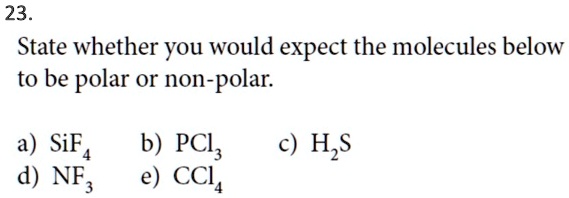
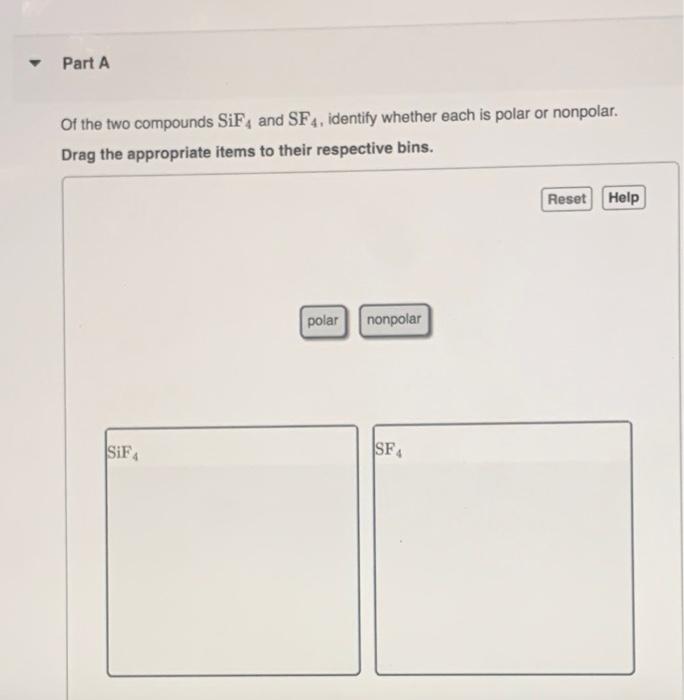
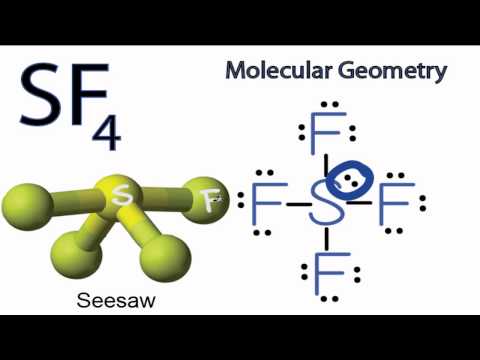
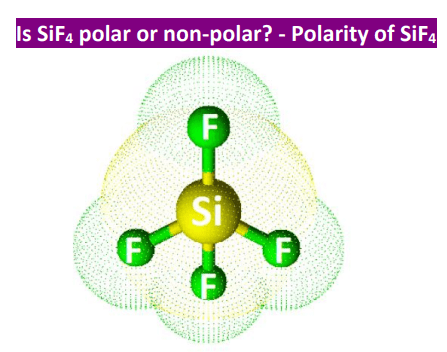
SOLVED: 23 State whether you would expect the molecules below to be polar or non-polar: a) SiF4 d) NF, b) PCl; e) CCL H,S

For SiF4, draw the Lewis structure, predict the shape, and determine if the molecule is polar or nonpolar. | Homework.Study.com