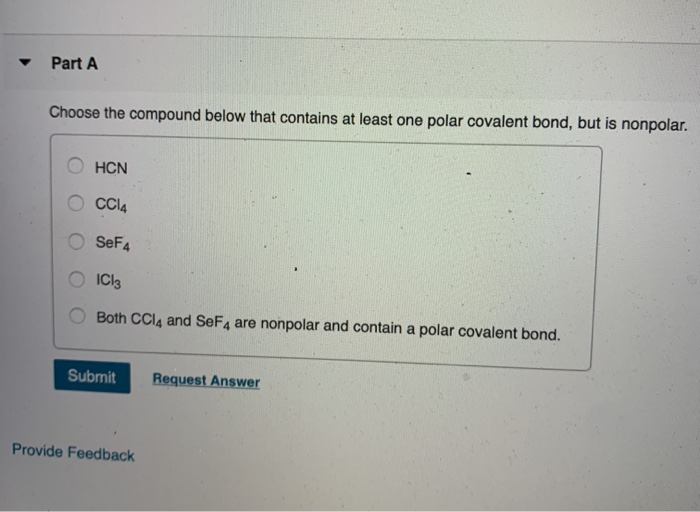
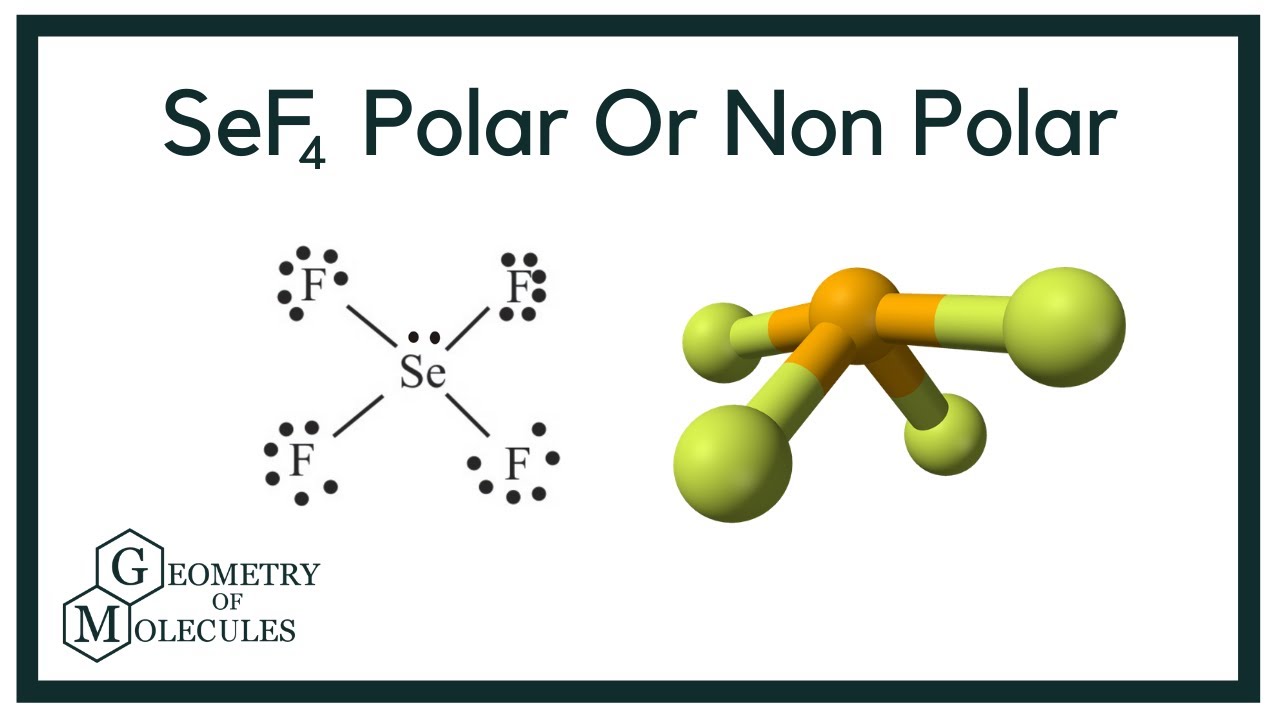
Consider the following statements and determine which are true and which are false. SeF4 is a polar molecule. NO3- has only two resonance Lewis structures. Bond angles for IF6+ are 60 .

SOLVED: Molecular Structure and Physical Properties Consider the following statements and determine which are true and which are false Bond angles for IF6 are 60 SeF4 has see-saw structure NO3 has only

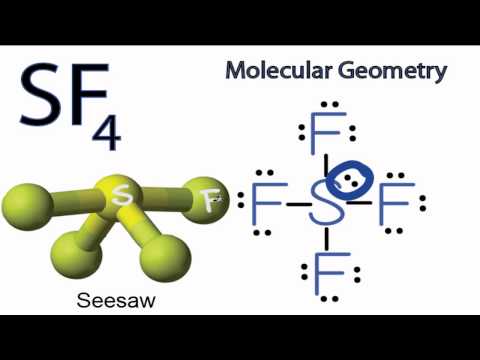
Consider the molecule SeF4. a. Draw the Lewis structure. b. What is the hybridization of Se? c. What is the electron geometry? d. What is the molecular geometry? e. What degree angles

SOLVED: match each of these molecules with the Polarity of the Molecule ( Polar/Nonpolar) CH2F2 SeH31+ SbF61- IBr41- TeF31- SeF4 BiCl5