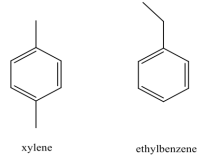
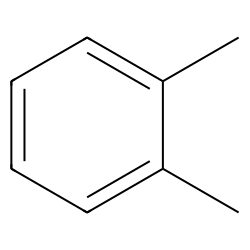
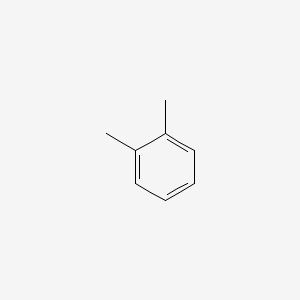
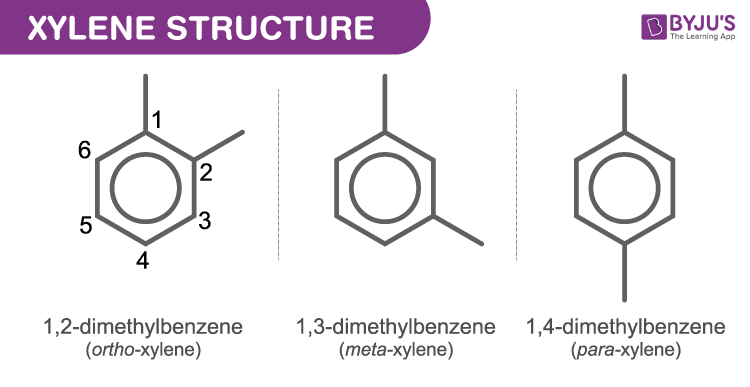
Chlorination of o-xylene (o-dimethylbenzene) yields a mixture of two products, but chlorination of p-xylene yields a single product. Explain. | Homework.Study.com

SOLVED: Was the compound in part oxidized or reduced? Explain your answcr: (4pts) The following compound is known as xylene: Is this compound polar or nonpolar? Explain your answer: In which compound

Separation of Xylene Isomers: A Review of Recent Advances in Materials | Industrial & Engineering Chemistry Research