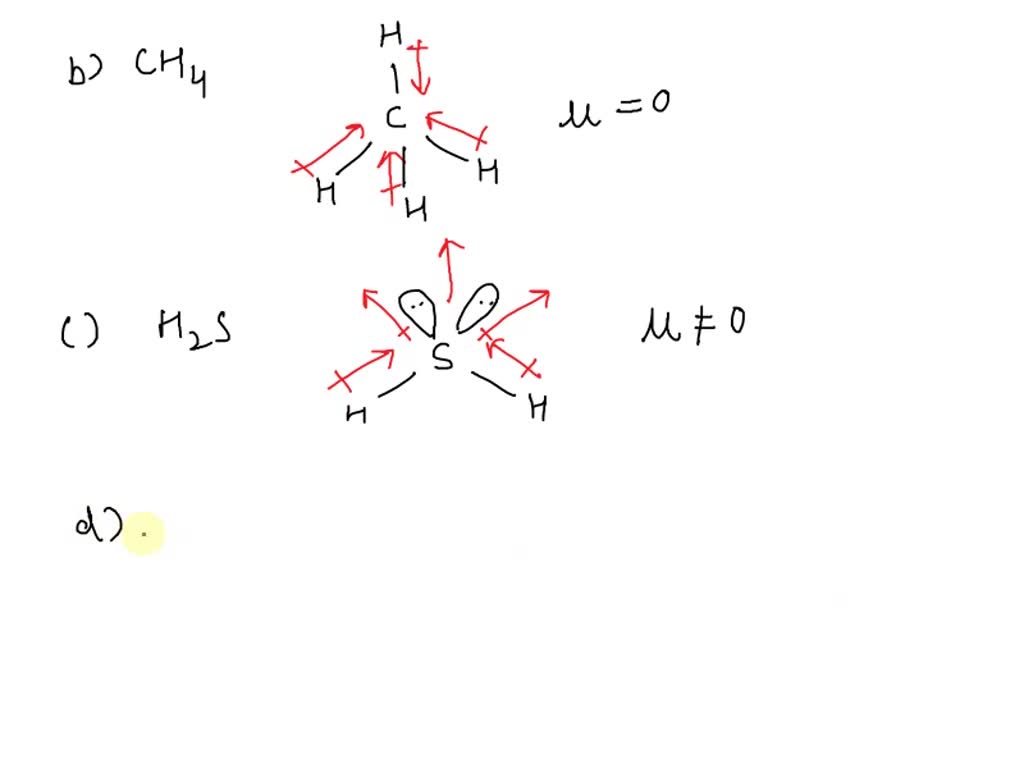
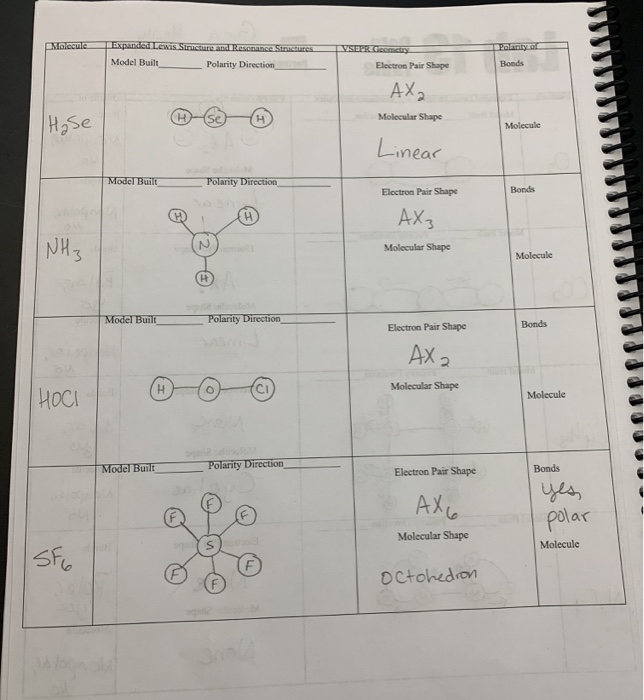
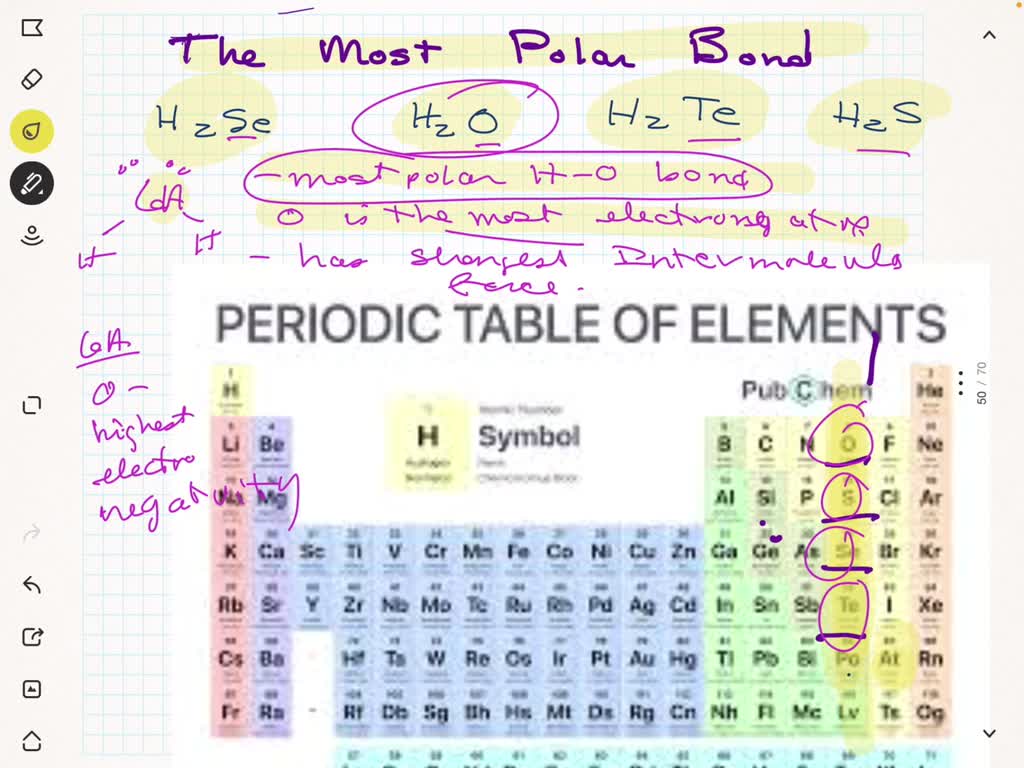
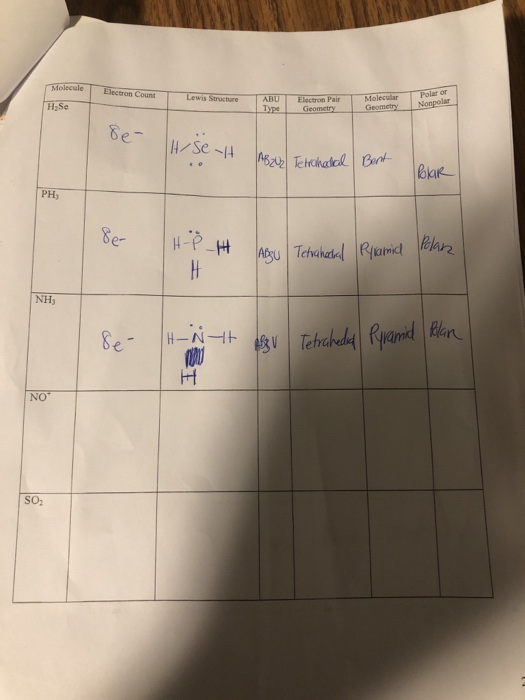
Warm-up For the following molecules, draw the Lewis diagram, determine the shape, and decide if it's polar or non-polar. H2Se CO2 CF4 CH2F2. - ppt download
Chemistry 1110 Covalent Properties I. Assign electronegativity values to the following elements: N I S P H B O F Br C II. Tell