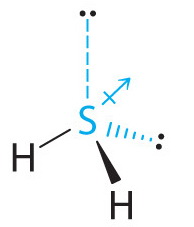
Explain the difference in the solubility of water and hydrogen sulfide in hexane? Solubility of water in hexane is 0.01 g/100g Solubility of hydrogen sulfide in hexane is 0.7g/100g? | Socratic

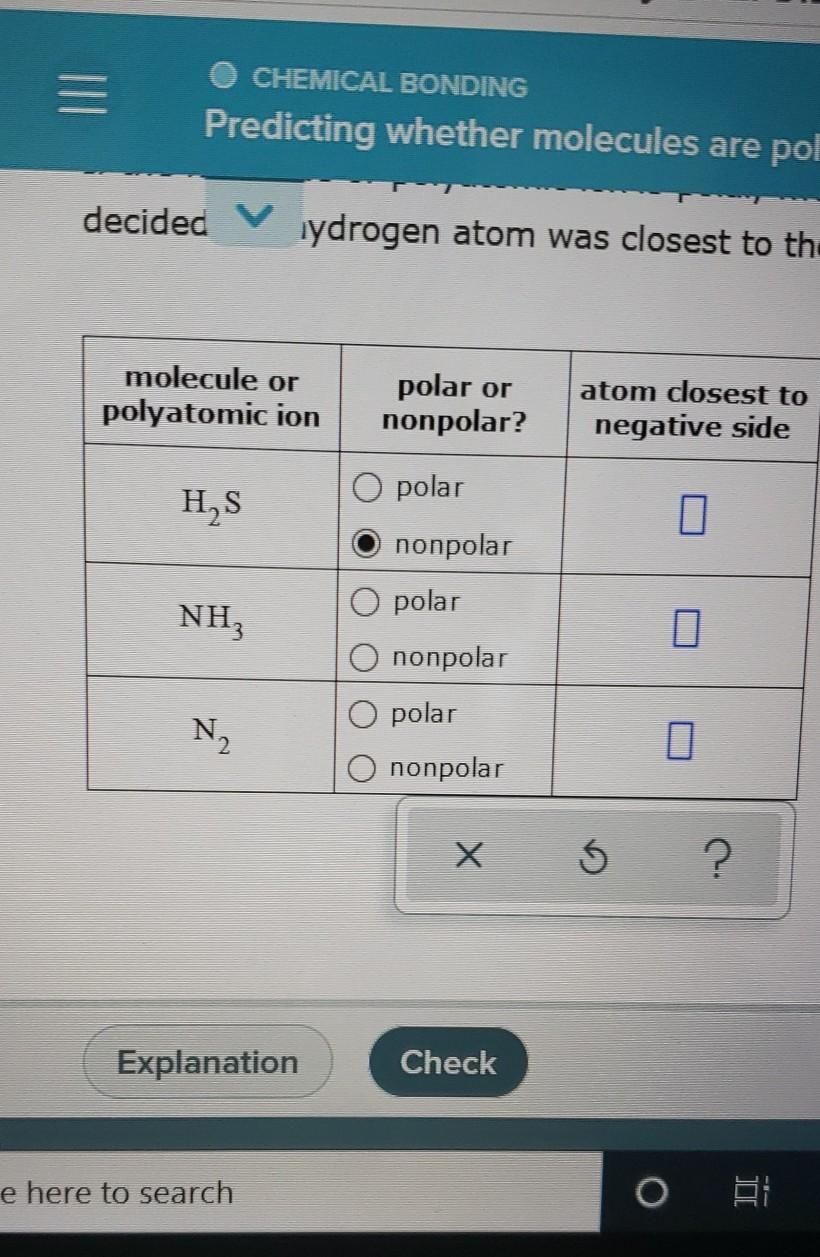
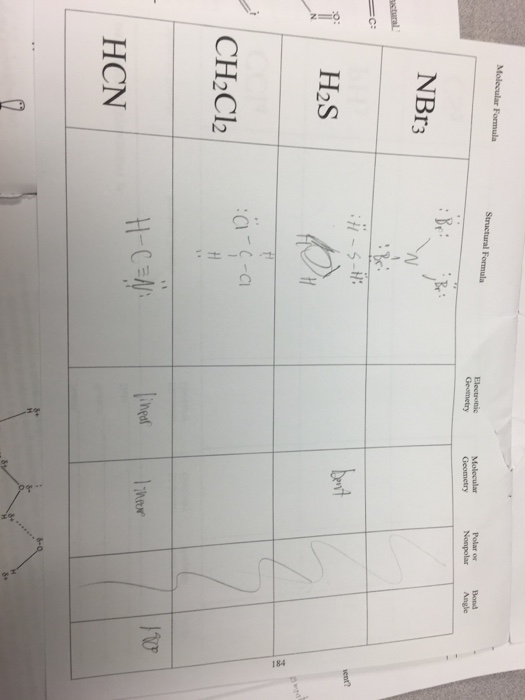
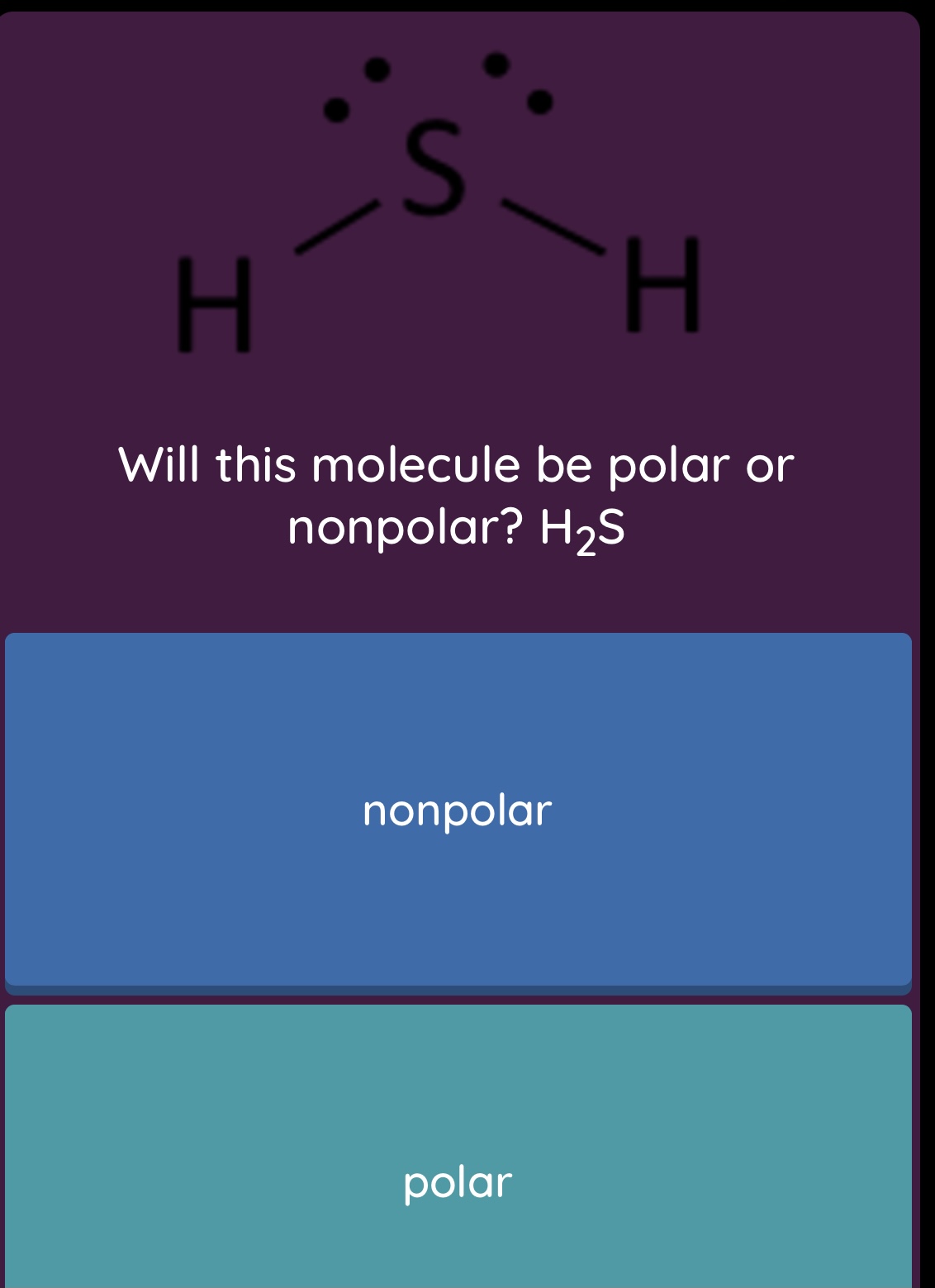
SOLVED: molecule or polyatomic ion polar or atom closest to nonpolar? negative side polar nonpolar polar nonpolar polar nonpolar H2S CH;F Cl2
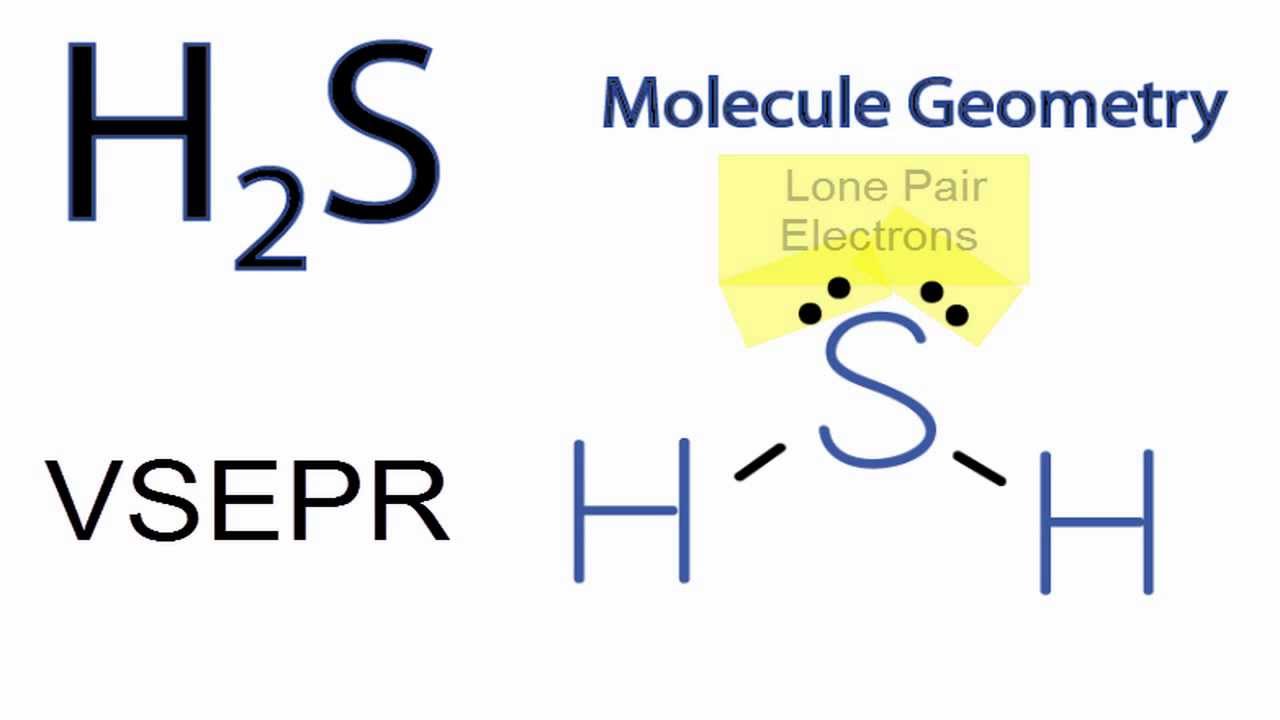
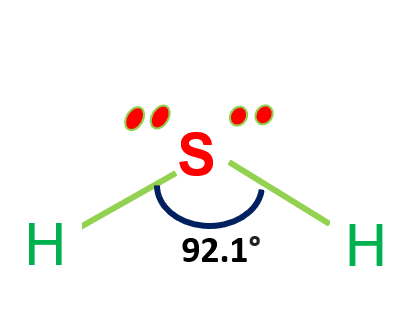
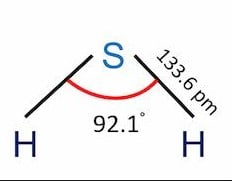
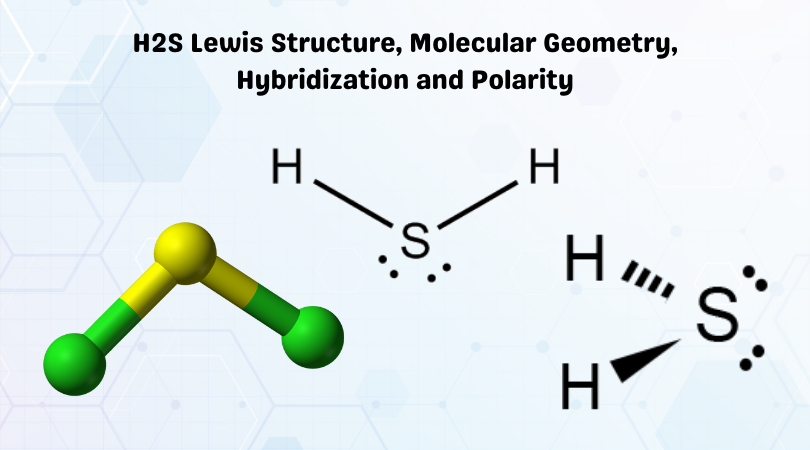
H2S Molecular Geometry / Shape and Bond Angles (Note: precise bond angle is 92.1 degrees.) - YouTube






![Is [math]H_2S[/math] polar or nonpolar? - Quora Is [math]H_2S[/math] polar or nonpolar? - Quora](https://qph.cf2.quoracdn.net/main-qimg-8f29d6cf5c618a0b64857748972fa0f3.webp)