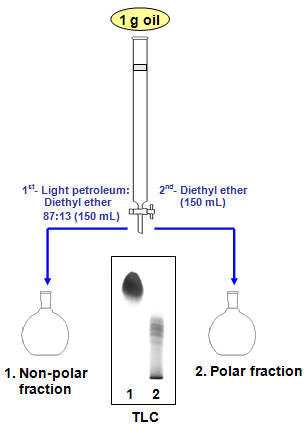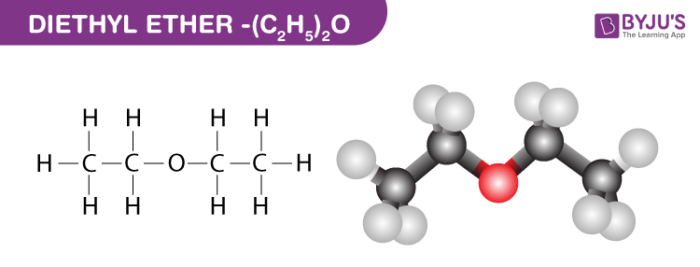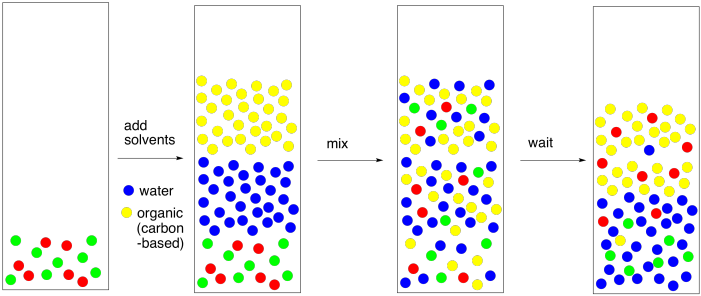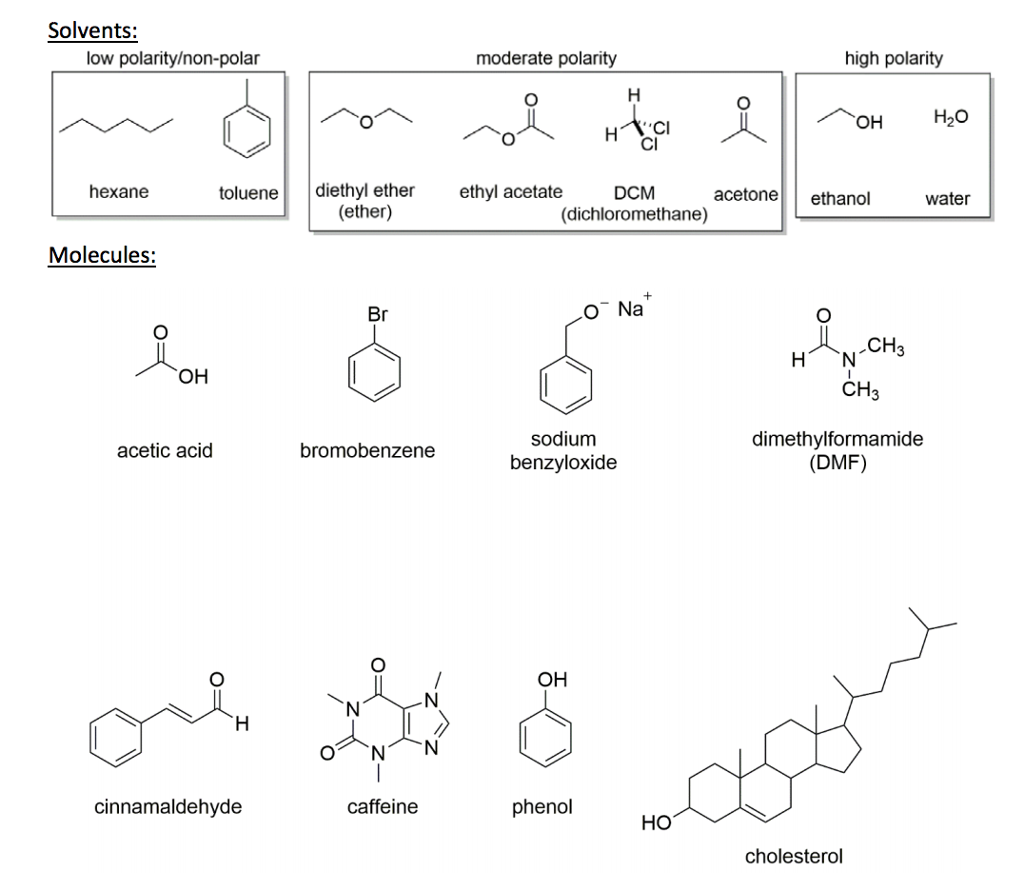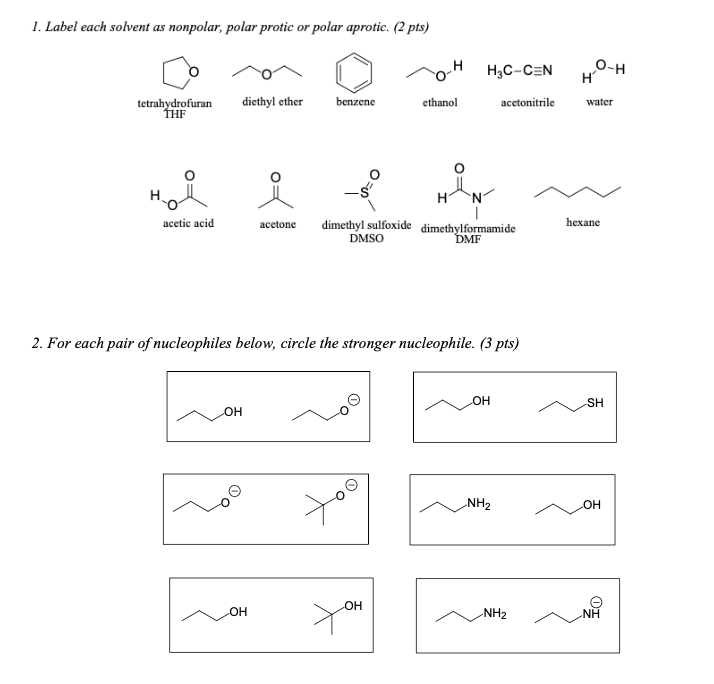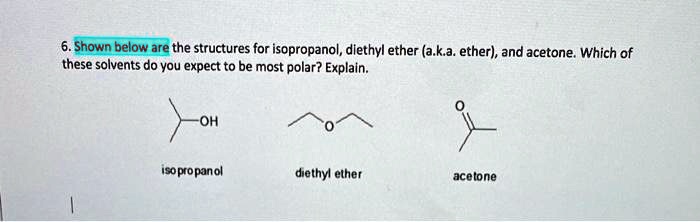
SOLVED: Shown below are the structures for isopropanol, diethyl ether (a.k.a. ether), ad acetone: Which of these solvents do vou expect to be most polar? Explaln; OH Isopropanol diethyl ether acetone

How can benzoic acid dissolve into diethyl ether since benzoic acid is polar and diethyl ether is non-polar? | Homework.Study.com

State, whether the following statements are True or False: Ethers are more polar than the isomeric alcohols.

Diethyl ether has a much higher boiling point than butane despite having a higher molecular weight. Explain why this is the case, making reference to the molecular structures of both compounds.